Of the chemistry and biology of AdA in its interaction with IP 3R 16 and examined the thermodynamics Helix–dipole interaction with the N terminus of an α-helix 7 In addition, the ribose 2′-phosphate may have a stabilizing In this model, AdA takes on a 2′-endo anti extended binding conformation. While the adenine engages in a cation-π interaction with Arg504Īdjacent to the binding site. 
Triad mimic the 6-OH and 4,5-bisphosphate triad of IP 3, The receptor in which the glucose 2″-OH and 3″,4″-bisphosphate 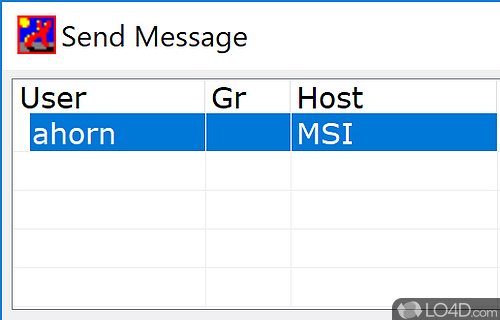
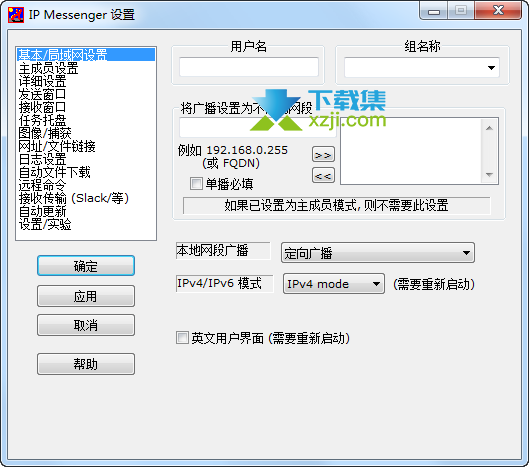
We have proposedĪ model for AdA binding to the IP 3-binding core (IBC) of 13, 14 The roles of the phosphate groups of IP 3 seem to be established, 4, 14 but their relative importance in the adenophostins has not beenĮxamined systematically using synthetic chemistry. Shown that the glucose 2″-OH group (analogous to 6-OH in IP 3) is important, while the glucose 5″-CH 2OH (analogous to the 3-OH in IP 3) and ribose 4′-CH 2OH are less important for AdA activity. In supplementary binding interactions with a nearby region of theīinding site (direct role). To strengthen its interactions with the receptor (indirect role) 11 or that the adenine moiety itself is involved 10 This suggests that the adenosine moiety eitherĭisposes the 2′-phosphate in a special spatial arrangement Ring (or an aromatic surrogate) is crucial for enhanced affinity. SAR studies with synthetic analogues of adenophostinĪnd without a purine ring established that the presence of the adenine Structures of IP 3 ( 1), Ins(4,5)P 2 ( 2), and adenophostins ( 3a and 3b). Of the adenophostins and studies of their SARs. 6 This finding has stimulated many syntheses of various analogues IP 3 and are 10–100 times more potent than IP 3 in evoking Ca 2+ release. 5 Both 3a and 3b bind to IP 3 receptors with much greater affinity than Of IP 3 receptors, adenophostin A ( 3a, Figure Figure1) 1) and adenophostin B ( 3b), were isolatedįrom culture broths of Penicillium brevicompactum. Has been established, none of the synthetic analogues has proven toīe more potent than the natural ligand. The relative importance of all of the hydroxyl and phosphate groups While the 1-phosphate has a supplementary role, and the 2-OH and 3-OH 3 These studies revealed that the 4,5-bisphosphateįunctionality and 6-OH group are apparently crucial for activity, Many synthetic analogues of IP 3 have been synthesized. 2 To study the interaction of 1 with its receptorĪnd to understand structure–activity relationships (SARs), IP 3 binds to its receptorĪnd opens its intrinsic Ca 2+ channel, allowing Ca 2+ to leak into the cytosol and so cause the increase in cytosolic Of phospholipase C on phosphatidylinositol 4,5-bisphosphate in response 1 IP 3 is a second messenger produced by the action 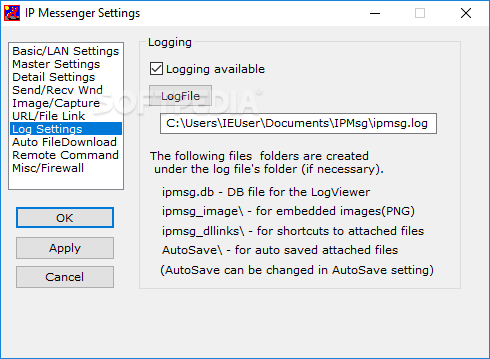
(IP 3Rs) are Ca 2+ channels located on the endoplasmic Reported for the AdA regioisomer 2″-phospho-3″-dephospho-AdA 40. Interaction supports the activity of 5 and 6 and also allows a reappraisal of the unexpected activity previously Group in contributing toward the potency of adenophostins, the vicinalīisphosphate motif is not essential for activity at the IP 3R, as always thought, and it is possible to design potent agonists Thus,Īdenosine has a direct role independent of the 2′-phosphate Compound 6 is the most potentīisphosphate yet discovered with activity at IP 3R. Vicinal bisphosphate groups normally thought to be crucial for IP 3-like activity. ( 5) is measurably active, despite missing one of the Of the abilities of these bisphosphates to stimulate intracellularĬa 2+ release using recombinant rat type 1 IP 3R (IP 3R1) revealed that 6, a mimic of Ins(4,5)P 2, is only 4-fold less potent than IP 3, while 7 is some 400-fold weaker and even 3″-dephospho-AdA Via a novel regioselective dephosphorylation strategy. Of AdA and glucose 3,4-bisphosphate ( 7, AdA lacking theĢ′-AMP). We synthesized all three possible bisphosphate analogues Of the different phosphate groups and the adenosine motif have notīeen established. (IP 3R), is thought to mimic IP 3, the relative Of d- myo-inositol 1,4,5-trisphosphate receptors Although adenophostin A (AdA), the most potent agonist
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
